Global Vaccination papers as movement licenses, & NIH patented mRNA therapies- a plan, long in the making
EU Vaccine hesitancy surveys from 2018, Vaccine development & patent timelines, dating back many years
In politics, nothing happens by accident. If it happens, you can bet it was planned that way.
-Franklin D. Roosevelt.
What happens to freedom if medical therapies become compulsory?
Long before the current pandemic, the European Union had been in the planning stages of developing a “common vaccination card/passport.” This universal vaccination passport is also designed for the “Greater good” and will be implemented as a requirement to protect “Public health.” How might your liberty and livelihood be restricted if you do not submit to all the vaccines required for the universal passport? Populations around the world have been conditioned to think that everyone is a biological source of contamination — innately guilty of spreading disease. Showing no authorization of immunity, every person is to be considered a filthy vector of disease and is to be avoided at all costs. The vaccination passport is being presented as the ultimate solution to quell these disease fears. By showing electronic proof of vaccination, people will be able to “return to normal” – a term used by authorities to coerce people to believe that medical therapies (vaccines) are the passport to prove absolute immunization. The commercialization of Masking measures will continue until all citizens can prove they have received the authorized injections. The European Union has been working on global vaccine passports since 2018. The planning stages began in 2018 when the European Union released a survey comparing public confidence/skepticism toward vaccines. The survey, titled “2018 State of Vaccine Confidence” was presented to the European Commission.
The report then already used terms such as “vaccine hesitancy” and “vaccine confidence,” in an attempt to build public trust in the effectiveness and safety of vaccines. The report immediately blames “vaccine delays and refusals” as the sole reason why outbreaks occur, why people are dying. One of the ways these central vaccine planners can eliminate “vaccine delays and refusals” is to blame people for diseases they don’t have. The next step is to socially isolate anyone who refuses vaccines by restricting their liberties. A “common vaccine passport” achieves these goals. This passport is blatant coercion, a violation of informed consent in medicine, an attack on personal choice, and ultimately sets the stage for segregation of healthy, unvaccinated people from society.
The central vaccine planners do not care how they ultimately coerce or manipulate people into submission. Soon after this coercive report was published, the EU commissioned a technical report titled “Designing and implementing an immunization information system” which mapped a monitoring system to ensure compliance to vaccinations. The report examined the development of a “common vaccination card/ passport for all including a comprehensive electronic immunization information system that can monitor vaccine compliance. Finally, the report sought to “overcome the legal and technical barriers impeding the interoperability of national immunization information systems.” By the third quarter of 2019, the EU created the “Vaccination Roadmap” designed to combat “vaccine hesitancy.” During this time, social media platforms began to label any information about vaccine choice, natural immunity, vaccine risk, and vaccine injury as dangerous “misinformation.” Global Vaccination Summit and the “call to action” to implement the vaccine passport By September 2019, the EU and the World Health Organization held the “Global Vaccination Summit.” There, “influential” leaders announced “10 Actions towards Vaccination for All” – a coercive plan to manipulate people into vaccine compliance. To make matters worse, just a month later, Event 201 was held. Sponsored by Johns Hopkins Center for Health Security, the World Economic Forum, and the Bill & Melinda Gates Foundation, Event 201 simulated a pandemic exercise focusing on a zoonotic novel coronavirus originating in bats. By November of 2019, these central vaccine planners had suggested a “call to action.” All this planning came before the current pandemic. The people who have planned for a vaccine passport are now calling for a “certificate of immunity.” Central vaccine planners like Bill Gates are now parading the media, and are putting their “call to action” into motion. People are now being told by their governments and their media that life cannot go back to normal unless there is a vaccine.
Medical therapies/(“Vaccine“) development timeline
Most of the research work here happened from 2013- 2019. Everything after 2020 seems to have been just finalization.
Let’s take a look at the patent timelines:
NIH-Moderna Agreements:
There are two agreements that appear relevant to a COVID-19 vaccine candidate. Both had some information redacted.
2017-1179” In May 2019, NIH and Moderna entered into a “research collaboration agreement” to develop vaccine candidates against “Middle East Respiratory Syndrome” coronavirus (MERS-CoV) and Nipah virus. The project was focused on evaluating candidates in animal models. NIH signed an amendment to the document on January 13, 2020—the day the agency and Moderna finalized the design of their novel coronavirus vaccine. The amendment is significantly redacted and does not mention the new coronavirus. However, the close proximity suggests the terms of the contract may have been expanded to apply to this new project. At the very least, the terms may have been instructive for the mRNA-1273 project. The document notes that ownership of inventions in the performance of the research project will “follow inventorship” in accordance with U.S. law.
One of the signers in these documents, Barney Graham, is the Deputy Director of the Vaccine Research Center, at the NIAID. https://www.niaid.nih.gov/research/barney-graham-md-phd
His accomplishments to date, are listed on his official profile page of the institute.
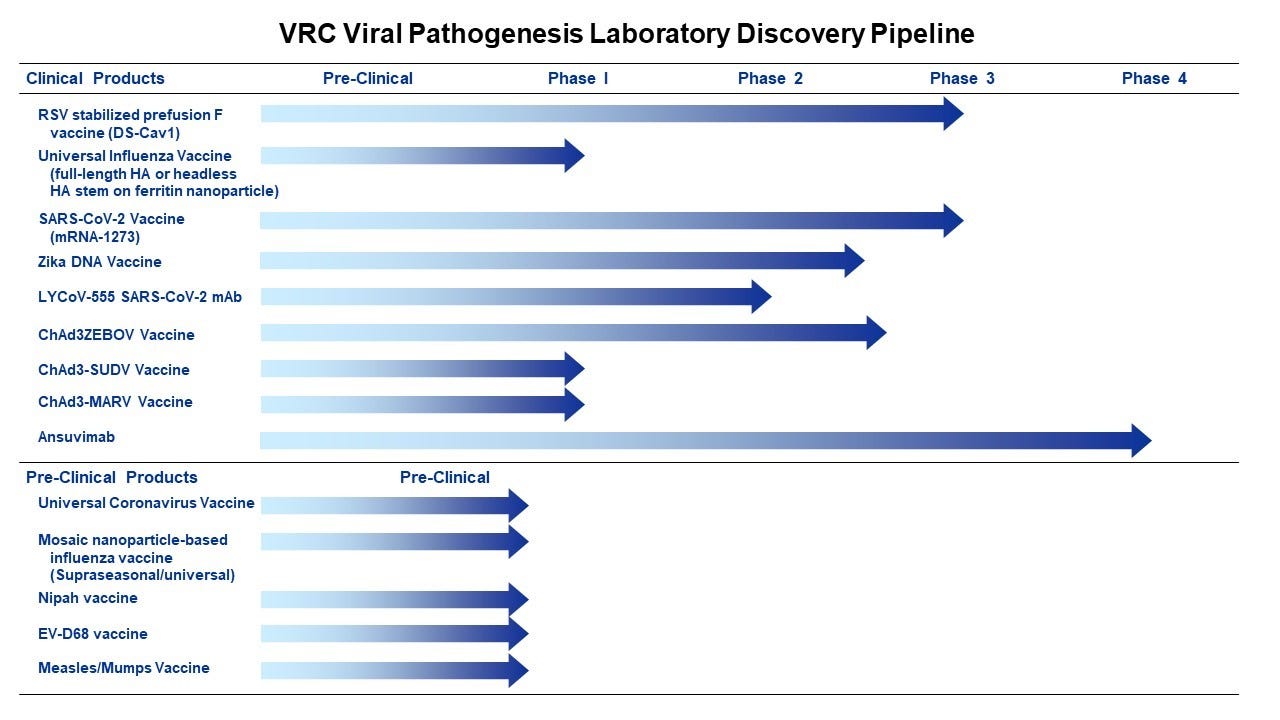
Interestingly, some of the main areas of research listed in the research agreements above, are still unlicensed and pre-clinical stage to this day. There is no licensed HIV vaccine, and Nipah Vaccine here is showing as pre-clinical.
So what was all that research, and the hush-hush agreements really for then?
Two patent application disclosures by federal scientists that appear relevant to a COVID-19 vaccine candidate. One application is provisional and remains unpublished. The other is nonprovisional and has been published. Provisional applications are easier to file and represent an initial step towards seeking patent protection. Nonprovisional applications serve as the basis for patent examination.
“2019-nCoV Vaccine.”
U.S. Application no. 62/972,886
A provisional application has been filed for a patent that appears to claim a component of the vaccine. From publicly available documents, it appears that the list of inventors at least includes several federal scientists, along with academic researchers based at the University of Texas, Austin. Notably, the scientific paper that disclosed this patent included Moderna scientists, but they were not named as co-inventors. If the patent is granted and covers the vaccine, then the NIH has an ownership stake.
“Prefusion Coronavirus Spike Proteins and Their Use.
U.S. Application No. 16/344,774
In 2016, federal scientists in partnership with academic researchers developed a new way to stabilize coronavirus spike proteins. The approach required substituting two amino acids, known as prolines, between the central helix and heptad repeat(“the 2P approach”). The stabilized spike protein for an earlier coronavirus produced a stronger immune response at lower doses than the naturally occurring protein. The scientists filed a patent application. The patent application, in relevant part, claims:
Stabilized proteins produced using the 2P approach across a group of coronaviruses (Claims 1 and 5);
Nucleic acid molecules (e.g., RNA) encoding those proteins (Claims 39-41); and
Methods for generating an immune response and inhibiting infection with the coronavirus (Claims 45-49).
Moderna describes mRNA-1273 as an “mRNA vaccine against SARS-CoV-2 encoding for a prefusion stabilized form of the Spike (S) protein.” The vaccine uses the 2P approach to produce stabilized spike proteins. The patent owners could eliminate Moderna from using the invention. The application lists numerous inventors at the National Institutes of Health, along with other researchers. The application states that it is owned by the U.S. government, Dartmouth College, and the Scripps Research Institute. If the patent is granted, then the NIH likely has an ownership stake.
NIH and Moderna agreed to transfer materials to a coronavirus scientist at the University of North Carolina for animal testing in an agreement accomplished December 16, 2019. The materials were described as “mRNA coronavirus vaccine candidates developed and jointly-owned by [NIH] and Moderna.” This likely refers to work done on an earlier coronavirus, MERS-CoV. It nonetheless provides further evidence that the federal government was developing coronavirus candidates with Moderna that is jointly owned.
In addition to potentially sharing the same collaboration agreement, the nature of the MERS and new coronavirus collaborations appear similar. In both cases, the NIH worked with Moderna to develop a coronavirus vaccine that expressed a stabilized spike protein.
Co-ownership provides significant rights. For example, assuming there were no other monopolies, the government could make, use, or sell the technology without the consent of Moderna. It could also license the technology to others, including other vaccine manufacturers or the World Health Organization, without the consent of Moderna.
The U.S. government provided millions of dollars to Moderna as early as 2013 to help develop its mRNA technology. The NIH meanwhile was also developing new methods to target coronavirus spike proteins.
Sources:
https://www.niaid.nih.gov/
https://s3.documentcloud.org/documents/6935295/NIH-Moderna-Confidential-Agreements.pdf
https://ec.europa.eu/health/sites/default/files/vaccination/docs/2018_vaccine_confidence_en.pdf










Good stuff. These players have been up to no good for well over 15 years.